Kinetochore oscillations in mitotic metaphase
During mitosis, sister chromatids connect to microtubules extending from the opposite spindle poles via kinetochores, protein complexes on the chromosome. When a pair of sister kinetochores establishes this configuration, they oscillate around the equatorial plane of the spindle, and the amplitude of these oscillations depends on kinesin-8 motors. During the oscillations sister kinetochores are separated due to tension, which has been proposed to regulate the spindle assembly checkpoint. The aim of this project is to study the mechanism of kinetochore oscillations.
Research area: Cell biology
Researcher: prof.dr.sc. Iva Tolić
Host Institution: Ruđer Bošković Institute, Zagreb
Project: Kinetochore oscillations in mitotic metaphase
DFG funding: € 255,950 for the whole team
Featured publication
Metaphase kinetochore movements are regulated by kinesin-8 motors and microtubule dynamic instability.
Anna H. Klemm, Agneza Bosilj, Matko Glunčić, Nenad Pavin, Iva M. Tolić. Mol Biol Cell, 29(11): 1332-1345 (2018). PDF | web
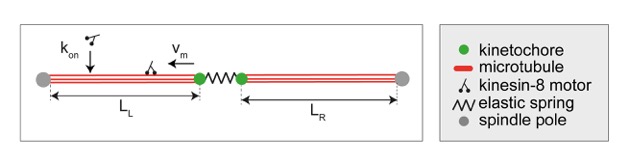
During metaphase, sister chromatids are connected to microtubules extending from the opposite spindle poles via kinetochores to protein complexes on the chromosome. Kinetochores congress to the equatorial plane of the spindle and oscillate around it, 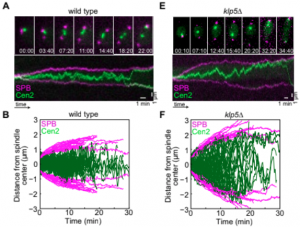 with kinesin-8 motors restricting these movements. Yet, the physical mechanism underlying kinetochore movements is unclear.We show that kinetochore movements in the fission yeast Schizosaccharomyces pombe are regulated by kinesin-8-promoted microtubule catastrophe, force-induced rescue, and microtubule dynamic instability. A candidate screen showed that among the selected motors only kinesin-8 motors Klp5/Klp6 are required for kinetochore centering. Kinesin-8 accumulates at the end of microtubules, where it promotes catastrophe. Laser ablation of the spindle resulted in kinetochore movement toward the intact spindle pole in wild-type and klp5Δ cells, suggesting that kinetochore movement is driven by pulling forces. Our theoretical model with Langevin description of microtubule dynamic instability shows that kinesin-8 motors are required for kinetochore centering, whereas sensitivity of rescue to force is necessary for the generation of oscillations. We found that irregular kinetochore movements occur for a broader range of parameters than regular oscillations. Thus, our work provides an explanation for how regulation of microtubule dynamic instability contributes to kinetochore congression and the accompanying movements around the spindle center.
with kinesin-8 motors restricting these movements. Yet, the physical mechanism underlying kinetochore movements is unclear.We show that kinetochore movements in the fission yeast Schizosaccharomyces pombe are regulated by kinesin-8-promoted microtubule catastrophe, force-induced rescue, and microtubule dynamic instability. A candidate screen showed that among the selected motors only kinesin-8 motors Klp5/Klp6 are required for kinetochore centering. Kinesin-8 accumulates at the end of microtubules, where it promotes catastrophe. Laser ablation of the spindle resulted in kinetochore movement toward the intact spindle pole in wild-type and klp5Δ cells, suggesting that kinetochore movement is driven by pulling forces. Our theoretical model with Langevin description of microtubule dynamic instability shows that kinesin-8 motors are required for kinetochore centering, whereas sensitivity of rescue to force is necessary for the generation of oscillations. We found that irregular kinetochore movements occur for a broader range of parameters than regular oscillations. Thus, our work provides an explanation for how regulation of microtubule dynamic instability contributes to kinetochore congression and the accompanying movements around the spindle center.
Other publications
Pivoting of microtubules driven by minus-end-directed motors leads to spindle assembly.
Lora Winters, Ivana Ban, Marcel Prelogović, Iana M. Kalinina, Nenad Pavin, Iva M. Tolić.
BMC Biology, 17: 42 (2019). PDF | web
Microtubule sliding within the bridging fiber pushes kinetochore fibers apart to segregate chromosomes.
Kruno Vukušić, Renata Buđa, Agneza Bosilj, Ana Milas, Nenad Pavin, Iva M. Tolić.
Dev Cell, 43(1): 11-23 (2017). PDF | web
Overlap microtubules link sister k-fibres and balance the forces on bi-oriented kinetochores.
Janko Kajtez, Anastasia Solomatina, Maja Novak, Bruno Polak, Kruno Vukušić, Jonas Rüdiger, Gheorghe Cojoc, Ana Milas, Ivana Šumanovac Šestak, Patrik Risteski, Federica Tavano, Anna H. Klemm, Emanuele Roscioli, Julie Welburn, Daniela Cimini, Matko Glunčić, Nenad Pavin, Iva M. Tolić.
Nat Commun, 7: 10298 (2016). PDF | web
Meiotic nuclear oscillations are necessary to avoid excessive chromosome associations.
Mariola R. Chacon, Petrina Delivani, Iva M. Tolić.
Cell Rep, 17: 1632–1645 (2016). PDF | web
Kinesin-8 Motors Improve Nuclear Centering by Promoting Microtubule Catastrophe.
Matko Glunčić, Nicola Maghelli, Alexander Krull, Vladimir Krstić, Damien Ramunno-Johnson, Nenad Pavin, Iva M. Tolić.
Phys Rev Lett, 114(7): 078103 (2015). PDF
